Abstract
-
Background
Multiple chemical sensitivity (MCS) lacks definitive biomarkers, making differential diagnosis reliant on exclusion and symptom reproducibility. In automobile repair shops, volatile organic compounds (VOCs) from cleaning agents pose risks not only to primary users but also to co-workers via indirect exposure. We report a case of MCS suspected to be triggered solely by bystander VOC exposure, highlighting the clinical significance of uncaptured peak exposures in Korea.
-
Case presentation
A 23-year-old mechanic experienced recurrent headaches, throat irritation, and nausea immediately after co-workers used aerosolized cleaners in a poorly ventilated workspace. Symptoms consistently improved when he was away from work. Routine clinical evaluations were unremarkable. Although the patient underwent surgery for chronic rhinosinusitis, the symptoms persisted specifically upon re-exposure, effectively ruling out structural otorhinolaryngologic causes. Mental health screening indicated only mild depressive symptoms, insufficient to account for the severity of the physical symptoms. The Quick Environmental Exposure and Sensitivity Inventory (QEESI) scores were in the "suggestive" range for MCS. While routine workplace monitoring (8-hour time-weighted average [TWA]) showed non-detectable solvent levels, the nature of aerosol application suggested uncaptured short-term peak exposures. Based on the distinct temporal relationship, the patient was placed under medical observation and assessed as fit for work with restrictions on solvent exposure.
-
Conclusions
This case highlights the role of indirect VOC exposure as a trigger for MCS symptoms even when routine TWA monitoring indicates minimal concentrations. The discrepancy likely stems from the brief peak exposures triggering central sensitization, underscoring the limitations of standard monitoring. Management strategies must focus on peak-exposure control and fitness-for-work assessments that mandate strict avoidance of organic solvents, regardless of average ambient concentrations.
-
Keywords: Multiple chemical sensitivity; Volatile organic compounds; Occupational exposure; Inhalation exposure
BACKGROUND
Multiple chemical sensitivity (MCS), also known as idiopathic environmental intolerance, is defined as a clinical syndrome in which recurrent, non-specific, multi-system symptoms—such as headache, fatigue, throat discomfort, and cognitive decline—occur after exposure to low concentrations of various chemicals well below standard toxicity limits. In early reports, it was characterized by reproducible symptoms elicited even with low-level exposures,
1 and consensus criteria proposed by Cullen and others
1,2 emphasize a presentation involving a chronic course, symptomatic on exposure to chemically unrelated substances, and improvement with avoidance.
The pathophysiology of MCS remains incompletely elucidated, though a multifactorial mechanism involving central sensitization, autonomic dysregulation, and oxidative stress has been suggested.
3,4 Despite epidemiological studies indicating a substantial population burden of chemical intolerance, MCS remains frequently underdiagnosed in clinical practice due to limited medical awareness and the complexity of its presentation.
5-8 In this context, the Quick Environmental Exposure and Sensitivity Inventory (QEESI) can be used as a supportive assessment tool. It provides a structured evaluation of chemical intolerance, symptom severity, and life impact, and prior studies have reported acceptable validity of the Korean version, supporting its use in Korean clinical settings.
5,9,10
From an occupational perspective, automobile maintenance is a high-risk industry that involves exposure to complex volatile organic compounds (VOCs), such as toluene and xylene, found in brake cleaners and degreasers. Crucially, studies indicate that spraying these agents generates short-term "peak exposures" that can trigger symptoms even among "bystander" workers who are not directly handling the chemicals.
11,12 However, standard industrial hygiene assessments typically rely on time-weighted average (TWA) concentrations, which often fail to capture these instantaneous peaks. Consequently, workers may suffer from neuro-irritative symptoms or central sensitization despite routine monitoring showing compliance with exposure limits.
13-16
While a case of MCS caused by ignition coal fumes has been reported in Korea,
17 occupational cases involving modern industrial solvents, particularly regarding indirect exposure in vehicle maintenance, have rarely been documented. Here, we report a case of a Korean automobile mechanic suspected of having MCS triggered by indirect exposure to aerosolized cleaners. We describe the diagnostic process using the validated Korean QEESI and discuss the implications of peak exposure control and fitness-for-work assessments in managing chemically intolerant workers.
CASE PRESENTATION
Occupational history and exposure assessment
The patient was a 23-year-old male nonsmoker who began working at an automobile maintenance company in July 2019. His primary duties involved general vehicle maintenance, including the replacement and assembly of engine parts. Although he was not a primary sprayer, he performed tasks in an open-plan workspace where co-workers frequently used aerosolized cleaners (e.g., 3M Car Choke & Carb Cleaner) to remove oil residues. Consequently, he was subjected to repeated indirect exposure to solvent mists and vapors. The safety data sheet (SDS) of the cleaning solutions listed toluene (27%–37%), xylene (10%–17%), ethylbenzene (10%–20%), and n-hexane (0.1%–1%) as major components.
According to a structured interview, the patient worked in a shared, open-floor service area exceeding 330 m² without partitioned booths. While the overall building was spacious, the patient’s immediate work zone comprised approximately 250 m3. Despite its size, the workshop functioned as an enclosed indoor environment with limited air movement; no local exhaust ventilation (LEV) was installed at spray-use points, and there was no dedicated mechanical dilution system. Vehicle entry shutters remained closed except during vehicle ingress and egress. Ventilation relied primarily on high clerestory windows (approximately 2.0–2.5 m above floor level) that were typically kept closed. While windows and doors were opened briefly when odors accumulated, perceived air exchange remained insufficient to clear chemical mists.
The patient reported that aerosolized cleaner spraying by co-workers occurred on average three days per week, with 3–4 events per day depending on task demand. Each event typically lasted 1–2 minutes, often occurring in repeated short bursts. The distance between the patient and the source of emission varied between 1 and 20 m. Even during distant spraying, odors were perceived to disperse across the entire floor over time. During these events, the patient frequently moved away from the source or stepped outside to mitigate symptoms. He explicitly confirmed that he never directly handled or sprayed the product himself.
Regarding respiratory protection, the patient reported using either a disposable particulate respirator (KF94) or a 3M particulate mask. While he self-reported a satisfactory fit and had received standard safety training, no formal quantitative fit test was performed. Crucially, these respirators lacked organic-vapor cartridges. Therefore, meaningful protection against the gaseous VOCs identified in the SDS was likely negligible, facilitating the progression of chemical sensitization despite his adherence to perceived safety protocols.
Clinical course and differential diagnosis
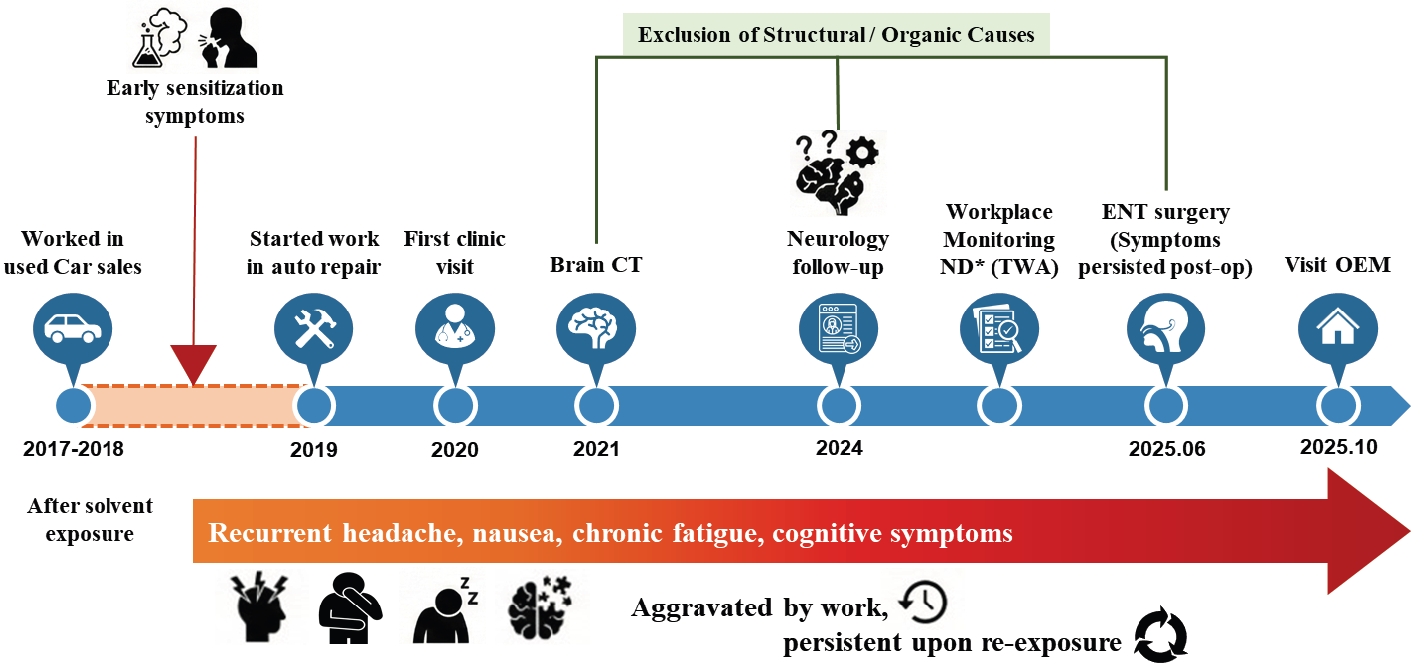
The clinical progression and diagnostic timeline are illustrated in
Fig. 1. Since commencing his duties in July 2019, the patient experienced intermittent headaches, throat irritation, nausea, and fatigue, typically emerging within minutes of co-workers spraying cleaning agents. These symptoms spontaneously resolved within several hours upon leaving the workplace or seeking fresh air. Retrospective inquiry revealed a history of mild dizziness and discomfort upon exposure to cleaning agents during previous employment in used-car sales (2017–2018). This history suggested a potential pre-existing susceptibility, significantly exacerbated by the poorly ventilated environment and repeated indirect exposures in his current job. Similar symptoms were reproduced at home upon exposure to household chemical odors, such as bleach or air fresheners.
In 2021, as the patient's symptoms began to interfere with his work performance, he sought medical evaluation. Brain computed tomography (CT) scans repeated over 3 years (July 2021 and August 2024) revealed no intracranial abnormalities. In the periodic special health examination conducted on November 6, 2024, laboratory findings—including complete blood count (CBC), standard serum biochemistry, and biological exposure indices for toluene (urinary o-cresol) and xylene (urinary methylhippuric acid)—were within normal limits. Chest radiography and pulmonary function tests were also unremarkable. Despite the absence of objective abnormalities and non-detectable levels of hazardous substances in routine workplace monitoring (8-hour TWA sampling), the occupational physician identified him as having suspected work-related neurologic symptoms requiring clinical follow-up. Consequently, he was assigned a conditional fitness-for-work status requiring strict exposure restrictions, which prioritized the avoidance of solvent exposure based on his clinical presentation.
In May 2025, the patient visited an otorhinolaryngology clinic for nasal obstruction and sneezing. Paranasal sinus CT revealed a deviated nasal septum, inferior turbinate hypertrophy, and chronic rhinosinusitis. He underwent revision endoscopic sinus surgery with septoturbinoplasty in June 2025. Although structural symptoms such as nasal obstruction improved post-operatively, his non-specific systemic symptoms (headache, nausea, cognitive fatigue) persisted specifically upon re-exposure to solvent odors. Consequently, he was referred to the Department of Occupational and Environmental Medicine at our hospital in October 2025 for further evaluation of suspected chemical sensitivity.
Routine laboratory investigations—including CBC, inflammatory markers (erythrocyte sedimentation rate, high-sensitivity C-reactive protein), and nutritional status (serum vitamin B12, folate, vitamin D)—were all within reference ranges, ruling out infectious, systemic inflammatory, or nutritional causes for the patient's fatigue and cognitive symptoms. While total IgE was mildly elevated (>100 IU/mL), the multiple allergen simultaneous test (MAST) panel revealed sensitization only to specific food antigens (peach and apple), with no reaction to common inhalant allergens. This finding supported the exclusion of typical IgE-mediated occupational asthma or rhinitis. The Sino-Nasal Outcome Test-22 score was 15, consistent with mild residual symptoms following sinus surgery. Furthermore, psychometric screening revealed a Patient Health Questionnaire (PHQ)-9 score of 5 (mild depression), Generalized Anxiety Disorder (GAD)-7 score of 2 (no anxiety), and Somatic Symptom Scale (SSS)-8 score of 5 (low somatic burden). These results suggested that a primary psychiatric disorder is unlikely to account for the severity and reproducibility of his physical symptoms (
Table 1).
To objectively quantify the patient's sensitivity, the Korean version of the QEESI (K-QEESI) was administered (
Table 2). The K-QEESI consists of five subscales, each containing 10 items rated on a 0–10 scale (score range: 0–100 per subscale). The scores for the three main scales were as follows: chemical intolerance: 42; symptom severity: 45; and life impact: 25. These values exceeded the cut-off criteria for the "high-risk" group (chemical intolerance ≥40, symptom severity ≥20), placing him in the "Suggestive" category for MCS.
Item-specific analysis revealed that the highest sensitivity scores were recorded for "household cleaning agents" (10/10) and "paint/thinner" (7/10), which correspond directly to the solvents used at his workplace. In contrast, intolerance to foods or medications was negligible (score 0), highlighting the chemical-specific nature of his condition.
The K-QEESI also includes a “Masking Index” which identifies ongoing exposures or behaviors—such as tobacco use, medications, or constant background irritants—that may obscure or “mask” an individual’s underlying sensitivity patterns. In this case, the overall masking index was relatively low (22 out of 100), suggesting that the patient’s clinical profile was not significantly confounded by background factors. However, the patient scored 8 out of 10 on item Q50 (“I am not sensitive to changes in indoor air quality”), which may indicate habituation to persistent background odors in the workshop. Taken together with the history of rapid symptom onset following aerosol spraying, this profile is compatible with the interpretation that his symptoms are more strongly associated with intermittent VOC spikes than with steady, low-level background exposure.
Ethics statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Institutional Review Board Committee of the Pusan National University Yangsan Hospital (IRB No. 55-2025-142 and date of approval was 31 October 2025). The requirement for informed consent was waived by the Institutional Review Board due to the retrospective nature of the study and the use of de-identified data.
DISCUSSION AND CONCLUSION
This case suggests that symptoms suggestive of MCS may develop in a worker who was not the primary sprayer, following indirect (“bystander”) exposure to aerosolized VOCs. The patient's odor-triggered systemic symptoms—headache, throat discomfort, and nausea—were repeatedly elicited during cleaner use and resolved within several hours after avoidance. This repeated occurrence of the symptoms and relief with avoidance is consistent with the clinical characteristics of MCS described in early reports.
1,2 A similar pattern was observed in the patient’s daily life. He experienced the same symptoms when exposed to household irritant odors such as bleach, detergents, and fabric softeners, consistent with previous findings that low-level chemical exposures in everyday environments may trigger symptoms in susceptible individuals.
6,7 Thus, this case represents an example of combined exposure across occupational and non-occupational settings, demonstrating the ‘spreading phenomenon’ typical of MCS.
1,2,18
During the diagnostic work-up at our center, the MAST Allergy 108-panel test showed only mild positivity for peach (3+) and apple (1+), and total IgE was mildly elevated (>100 IU/mL). Nevertheless, specific IgE to common inhalant allergens was not detected (
Table 1). These findings suggest a non-immune chemical intolerance rather than IgE-mediated allergy.
19 Mental-health and somatization screening (PHQ-9, GAD-7, and SSS-8) did not indicate major psychiatric illness (
Table 1). To further differentiate the clinical presentation, migraine or vestibular migraine was considered less likely as the patient’s headaches lacked pulsatile characteristics and were not accompanied by photophobia or phonophobia. Occupational asthma or reactive airways dysfunction syndrome was also excluded given the absence of wheezing, episodic dyspnea, or objective abnormalities on routine pulmonary function testing; furthermore, no panic or hyperventilation phenomena were reported. Notably, the patient's autonomic and cognitive symptoms persisted specifically upon chemical re-exposure, even after successful endoscopic sinus surgery had resolved his structural nasal obstruction. This clinical course, combined with the absence of structural brain abnormalities, allowed us to exclude purely rhinologic causes (e.g., chronic sinusitis), fibromyalgia syndrome, chronic fatigue syndrome, and somatic symptom disorder. Functional gastrointestinal disorders were considered less likely because nausea occurred primarily in close temporal proximity to solvent odor exposure rather than as a chronic independent symptom cluster. Additional tests such as bronchial provocation testing (e.g., methacholine challenge) or laryngeal evaluation were not pursued because there was no recurrent wheeze/dyspnea or voice-related symptom complex, and the clinical presentation was dominated by reproducible odor-triggered systemic symptoms. Ultimately, the QEESI profile (42/45/25; “Suggestive”) provided structured support for suspected MCS and was consistent with previously proposed assessment frameworks.
9,10
A critical challenge in diagnosing occupational MCS is the discrepancy between symptom severity and measured exposure levels. In this case, as real-time VOC monitoring or short-term, task-aligned sampling was not available during the period of sensitization, exposure characterization relied on task-based reconstruction via a structured interview. This reconstruction detailed the frequency of spray events, duration, distance from the source, and specific ventilation constraints. The fact that routine workplace monitoring based on 8-hour TWA measurements showed non-detectable solvent levels highlights a significant limitation of TWA-based evaluation in intermittent spray processes. Supporting this view, previous studies have reported that, in automobile repair work, the use of aerosol brake cleaners can cause short-term peak increases in airborne VOC concentrations, including toluene and xylene, of several-fold over background levels.
11,12 Importantly, although these transient spikes appeared to trigger symptoms in this patient, similar short-term exposures in occupational settings may remain below levels generally associated with acute systemic toxicity (e.g., narcosis) in the general population. This pattern is consistent with the classical definition of MCS, which characterizes the condition as reproducible symptoms elicited by exposures at levels significantly below the threshold for toxic effects.
1
The patient’s QEESI profile and the reproducible relationship between solvent exposure and symptoms are compatible with mechanisms proposed for MCS, including central sensitization and autonomic dysregulation.
20,21 Evidence of central sensitization and hyperresponsiveness in MCS has been reported by functional magnetic resonance imaging and neurophysiologic testing,
22,23 and the pattern of indirect peak exposures in this case may be interpreted along similar mechanistic lines. While organic solvents like toluene and xylene are well-known neurotoxicants capable of causing cognitive decline and neuropathy at high doses,
13-16 the mechanism in this case appears distinct from direct toxic encephalopathy.
Instead, consistent with recent literature, repeated intermittent exposures may act as physiological stressors associated with central sensitization and oxidative metabolic imbalance.
3,4 Even without structural brain damage, functional connectivity changes between the olfactory cortex and the limbic system (amygdala and cingulate cortex) can heighten sensory and emotional responses to chemical stimuli, as shown in neuroimaging studies of patients with MCS.
18,24 Furthermore, chemosensory irritant pathways involving transient receptor potential (TRP) channels, particularly transient receptor potential ankyrin 1 (TRPA1) and transient receptor potential vanilloid 1 (TRPV1) in the airway epithelium, have been proposed as potential contributors to odor-related symptom amplification. Repeated VOC exposure may increase TRP-mediated responsiveness and may be associated with neurogenic irritation even at sub-toxic exposure levels.
3,18 Mast cell activation syndrome (MCAS) has also been proposed as a potential biological correlate of chemical intolerance, where chemical triggers induce mediator release that accounts for multisystemic symptoms.
3,25 However, in this patient, typical systemic allergic features (e.g., urticaria, flushing, or anaphylaxis) were absent. Furthermore, as MCAS was not initially suspected during the diagnostic work-up, specific biomarkers such as baseline tryptase or urinary mediators were not evaluated. Therefore, MCAS remains a theoretical consideration rather than a supported diagnosis in this case. Overall, an integrated central–peripheral framework may help interpret the patient’s severe autonomic and cognitive symptoms despite normal brain CT findings.
Recent reviews describe MCS as a multifactorial environmental sensitivity condition reported across diverse demographic and occupational groups.
3,4,18,26 Epidemiologic data indicate a non-negligible population burden: in the Danish DanFunD study (2021), the prevalence of chemical sensitivity was approximately 1.9% (188/9,656)
8; earlier U.S. population survey data estimated chemical hypersensitivity at 11.2% (with 2.5% medically diagnosed MCS)
27; and a Korean hospital-based study reported 16.4% in the very suggestive QEESI category.
5 These estimates should be interpreted cautiously because case definitions and sampling frames differ across studies. Nonetheless, they support the possibility that cumulative environmental exposures are relevant to MCS. While most epidemiologic evidence comes from daily-life environments, this case highlights the clinical importance of transient, task-related exposures in industrial workplaces such as automobile maintenance.
In Korea, an earlier case was reported wherein a patient was diagnosed with MCS after acute exposure to ignition coal fumes, characterized by persistent headache, fatigue, cognitive decline, and hypersensitivity to various odors;
17 in that report, broad hypersensitivity to low-level chemicals and functional impairment in daily life were key features, similar to our case. However, for that patient, acute high-concentration exposure to combustion gases during coal ignition served as a clear single trigger event, whereas in our case, the symptoms emerged from the prolonged accumulation of transient occupational peaks. Internationally, individual case reports of MCS have also been published.
28,29 These reports commonly describe persistent broad hypersensitivity to low-level chemicals after exposures related to pesticide use, indoor remodeling, or irritants in residential environments. However, these cases focus on environmental exposures in daily life, with limited quantitative information on occupational exposure levels. This case provides additional clinical evidence regarding the role of transient occupational exposures in the development of MCS, complementing existing literature that often focuses on acute toxic events or domestic triggers.
From a management perspective, avoidance of exposure, workplace improvements (engineering controls), cognitive behavioral therapy, exposure-based behavioral therapy, and lifestyle modification have been suggested as potential strategies. In particular, cognitive behavioral therapy and exposure-based approaches may help reduce avoidance behaviors and conditioned autonomic responses to chemical exposure. When combined with improvements in the work environment, they have been associated with better symptomatic and functional outcome.
26,30 In this case, a multi-level intervention was implemented at our department of occupational and environmental medicine in November 2025. This included administrative measures, such as reassigning the patient to a unit where exposure to cleaners could be avoided, and engineering recommendations, such as improving general ventilation and installing LEV. Additionally, the patient was advised on lifestyle modifications, including the avoidance of irritant detergents in the home. This integrated approach may serve as a practical model for managing workers with suspected MCS and for reducing detrimental occupational exposures.
This case report has several limitations. First, exposure assessment was retrospective and relied on routine 8-hour TWA monitoring and task-based reconstruction. Because direct real-time monitoring and short-term sampling during active spraying events were unavailable, the peak-exposure inference remains a clinical interpretation rather than a directly captured measurement. Second, MCS currently lacks definitive objective biomarkers, and although the clinical course and QEESI scores were suggestive, causal attribution in this case remains limited. Third, despite a structured differential diagnosis and psychiatric screening, the potential for residual confounding by unmeasured psychosocial factors or other environmental stressors cannot be excluded.
In summary, this case highlights practical implications for managing suspected chemical intolerance in vehicle maintenance settings. Prevention strategies in similar workplaces should prioritize respirators equipped with organic vapor cartridges rather than simple particulate masks, together with regular fit testing and worker training on the risks of indirect exposure. Departments of Occupational and Environmental Medicine can serve as hubs for clinical differentiation, environmental assessment, and follow-up to support the management of workers with suspected chemical intolerance.
Abbreviations
Generalized Anxiety Disorder-7
local exhaust ventilation
multiple allergen simultaneous test
mast cell activation syndrome
multiple chemical sensitivity
Patient Health Questionnaire-9
Quick Environmental Exposure and Sensitivity Inventory
transient receptor potential
transient receptor potential ankyrin 1
transient receptor potential vanilloid 1
volatile organic compound
NOTES
-
Funding
This study was supported by the 2023 overseas training grant from Pusan National University Yangsan Hospital.
-
Competing interests
No potential conflict of interest relevant to this article was reported.
-
Author contributions
Conceptualization: Kim SY. Data curation: Kim SY, Cheon DH. Methodology: Kim SY. Funding acquisition: Kim SY. Writing - original draft preparation: Kim SY, Cheon DH. Writing - review & editing: Kim SY, Kim YJ, Kang D, Kim Y, Lee ES, Kim KH.
Fig. 1.Clinical and occupational timeline illustrating symptom progression and diagnostic exclusion in a worker with suspected multiple chemical sensitivity. CT: computed tomography; ENT: ear, nose, and throat; OEM: Original Equipment Manufacturer; TWA: time-weighted average.
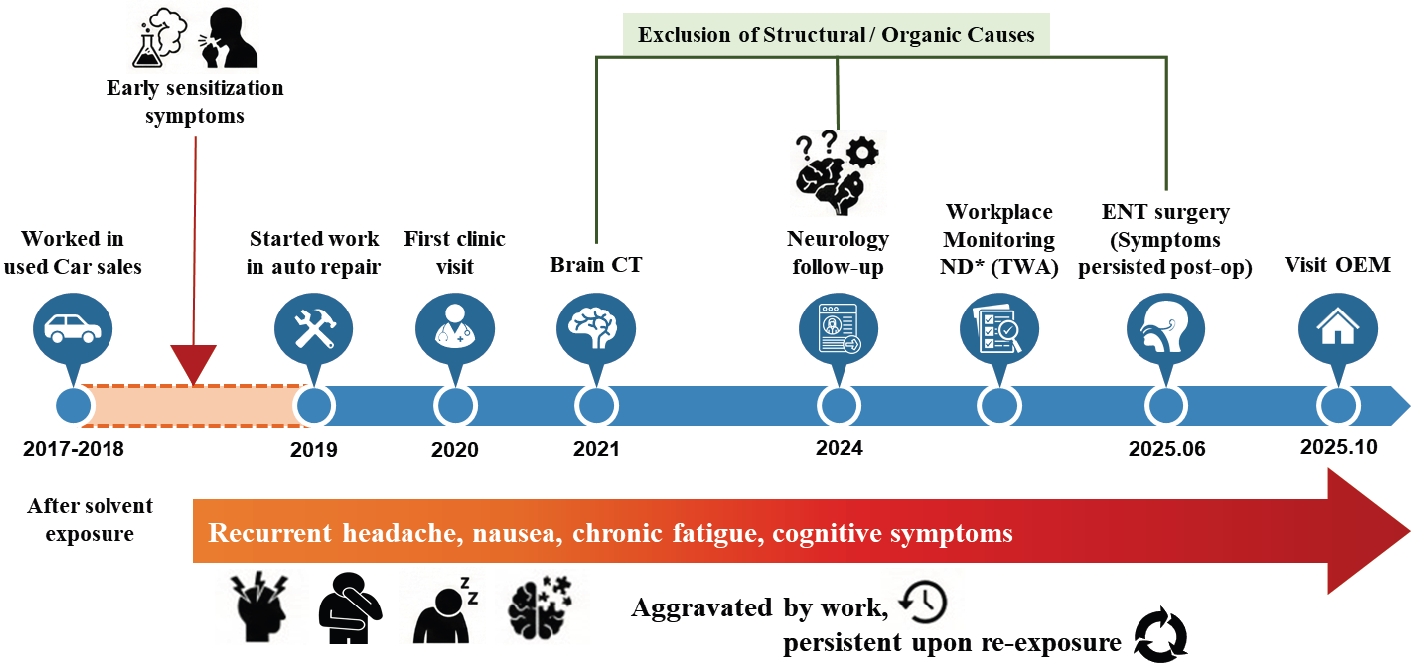
Table 1.Summary of clinical laboratory findings and psychometric assessments regarding exclusion diagnosis
|
Category |
Test items |
Unit |
Results |
Reference range |
Interpretation |
|
Hematology |
WBC |
10³/µL |
5.89 |
4.0–11.0 |
WNL |
|
Hemoglobin |
g/dL |
16.4 |
13.5–17.5 |
WNL |
|
Platelet count |
10³/µL |
299 |
140–400 |
WNL |
|
Eosinophils |
% |
1.0 |
0–6 |
No eosinophilia |
|
ESR |
mm/h |
2 |
0–10 |
No active inflammation |
|
Immunology |
hsCRP |
mg/dL |
0.07 |
0–0.5 |
|
|
Total IgE |
IU/mL |
>100 |
≤100 IU/mL |
Elevated |
|
MAST (108 items) |
|
Inhalant: negative; food: positive (peach 3+, apple 1+) |
- |
No respiratory allergy; elevated IgE due to food sensitization |
|
Nutritional |
Vitamin B12 |
pg/mL |
449 |
187–883 |
Normal |
|
Folate |
ng/mL |
5.6 |
3.1–20.5 |
Normal |
|
25-OH Vitamin D |
ng/mL |
16.65 |
6.6–49.9 |
Normal |
|
Psychometric |
PHQ-9 |
|
5 |
0–4 (normal) |
Mild depression |
|
GAD-7 |
|
2 |
0–4 (normal) |
Normal (no anxiety) |
|
SSS-8 |
|
5 |
0–3 (normal) |
Mild somatic burden |
|
Rhinology |
SNOT-22 |
|
15 |
0–7 (normal) |
Mild residual CRS symptoms |
Table 2.Results of the Korean version of the Quick Environmental Exposure and Sensitivity Inventory (QEESI)
|
QEESI scale |
Patient score (0–100) |
Risk criteria (Korean validation)a
|
Classification |
Key high-scoring items (item score 0–10) |
|
Chemical intolerance |
42 |
40–59 (suggestive) |
Suggestive |
Household cleaning agents (10) |
|
≥60 (very suggestive) |
Paint/thinner (7) |
|
Pesticides/insect sprays (5) |
|
Diesel/gasoline fumes (4) |
|
Symptom severity |
45 |
40–59 (suggestive) |
Suggestive |
Headache (10) |
|
≥60 (very suggestive) |
Shortness of breath (8) |
|
Fatigue (7) |
|
Insomnia (7) |
|
Concentration difficulty (5) |
|
Life impact |
25 |
20–39 (suggestive) |
Suggestive |
Ability to work or study (10) |
|
≥40 (very suggestive) |
Emotional control (7) |
|
Other intolerance |
7 |
- |
Low |
Mold, dust, or pollen (3) |
|
Bright lights (2) |
|
Emotional stress (2) |
|
Foods/medications: (0) |
|
Masking index |
22 |
- |
- |
"Not sensitive to indoor air changes" (8)b
|
|
Use of air conditioning (5) |
|
Drinking/smoking (3) |
REFERENCES
- 1. Cullen MR. The worker with multiple chemical sensitivities: an overview. Occup Med 1987;2(4):655–61.
- 2. Multiple chemical sensitivity: a 1999 consensus. Arch Environ Health 1999;54(3):147–9.ArticlePubMed
- 3. Lavric CE, Migueres N, de Blay F. Multiple chemical sensitivity: a review of its pathophysiology. Explor Asthma Allergy 2024;2:350–62.Article
- 4. Rossi S, Pitidis A. Multiple chemical sensitivity: review of the state of the art in epidemiology, diagnosis, and future perspectives. J Occup Environ Med 2018;60(2):138–46.PubMedPMC
- 5. Jeong I, Kim I, Park HJ, Roh J, Park JW, Lee JH. Allergic diseases and multiple chemical sensitivity in Korean adults. Allergy Asthma Immunol Res 2014;6(5):409–14.ArticlePubMedPMC
- 6. Suzuki T, Bai Y, Ohno Y. Prevalence and factors related to high risk of multiple chemical sensitivity among Japanese high school students. Int J Environ Res Public Health 2024;21(7):934.ArticlePubMedPMC
- 7. Caress SM, Steinemann AC. Prevalence of multiple chemical sensitivities: a population-based study in the southeastern United States. Am J Public Health 2004;94(5):746–7.ArticlePubMedPMC
- 8. Dantoft TM, Nordin S, Andersson L, Petersen MW, Skovbjerg S, Jorgensen T. Multiple chemical sensitivity described in the Danish general population: cohort characteristics and the importance of screening for functional somatic syndrome comorbidity-The DanFunD study. PLoS One 2021;16(2):e0246461.ArticlePubMedPMC
- 9. Miller CS, Prihoda TJ. The Environmental Exposure and Sensitivity Inventory (EESI): a standardized approach for measuring chemical intolerances for research and clinical applications. Toxicol Ind Health 1999;15(3-4):370–85.ArticlePubMed
- 10. Jeon BH, Lee SH, Kim HA. A validation of the Korean version of QEESI(c) (The Quick Environmental Exposure and Sensitivity Inventory). Ann Occup Environ Med 2012;24(1):96–114.
- 11. Fries M, Williams PR, Ovesen J, Maier A. Airborne exposures associated with the typical use of an aerosol brake cleaner during vehicle repair work. J Occup Environ Hyg 2018;15(7):531–40.ArticlePubMed
- 12. Wilson MP, Hammond SK, Nicas M, Hubbard AE. Worker exposure to volatile organic compounds in the vehicle repair industry. J Occup Environ Hyg 2007;4(5):301–10.ArticlePubMed
- 13. Dick FD. Solvent neurotoxicity. Occup Environ Med 2006;63(3):221–6.ArticlePubMedPMC
- 14. Niaz K, Bahadar H, Maqbool F, Abdollahi M. A review of environmental and occupational exposure to xylene and its health concerns. EXCLI J 2015;14:1167–86.PubMedPMC
- 15. Mitran E, Callender T, Orha B, Dragnea P, Botezatu G. Neurotoxicity associated with occupational exposure to acetone, methyl ethyl ketone, and cyclohexanone. Environ Res 1997;73(1-2):181–8.ArticlePubMed
- 16. El-Hagrasy AM, Karrout RH, Mcguinness AL, Albutain TZ, Khalifa D, Khalil FM, et al. Investigating the general effects of different types of toluene exposure on the health of workers: an integrative review of the literature. BMJ Public Health 2025;3(1):e001046.ArticlePubMedPMC
- 17. Yun MJ, Kang DM, Lee KH, Kim YK, Kim JE. Multiple chemical sensitivity caused by exposure to ignition coal fumes: a case report. Ann Occup Environ Med 2013;25(1):32.ArticlePubMedPMC
- 18. Jacques L. Multiple chemical sensitivity: a clinical perspective. Brain Sci 2024;14(12):1261.ArticlePubMedPMC
- 19. Ansotegui IJ, Melioli G, Canonica GW, Caraballo L, Villa E, Ebisawa M, et al. IgE allergy diagnostics and other relevant tests in allergy, a World Allergy Organization position paper. World Allergy Organ J 2020;13(2):100080.ArticlePubMedPMC
- 20. Azuma K, Uchiyama I, Tanigawa M, Bamba I, Azuma M, Takano H, et al. Chemical intolerance: involvement of brain function and networks after exposure to extrinsic stimuli perceived as hazardous. Environ Health Prev Med 2019;24(1):61.ArticlePubMedPMCPDF
- 21. Volcheck MM, Graham SM, Fleming KC, Mohabbat AB, Luedtke CA. Central sensitization, chronic pain, and other symptoms: better understanding, better management. Cleve Clin J Med 2023;90(4):245–54.ArticlePubMed
- 22. Hillert L, Jovanovic H, Ahs F, Savic I. Women with multiple chemical sensitivity have increased harm avoidance and reduced 5-HT(1A) receptor binding potential in the anterior cingulate and amygdala. PLoS One 2013;8(1):e54781.ArticlePubMedPMC
- 23. Miki T, Inoue Y, Miyajima E, Kudo Y, Tsunoda M, Kan S, et al. Enhanced brain images in the limbic system by functional magnetic resonance imaging (fMRI) during chemical exposures to patients with multiple chemical sensitivities. Kitasato Med J 2010;40:27–34.
- 24. Azuma K, Uchiyama I, Takano H, Tanigawa M, Azuma M, Bamba I, et al. Changes in cerebral blood flow during olfactory stimulation in patients with multiple chemical sensitivity: a multi-channel near-infrared spectroscopic study. PLoS One 2013;8(11):e80567.ArticlePubMedPMC
- 25. Miller CS, Palmer RF, Dempsey TT, Ashford NA, Afrin LB. Mast cell activation may explain many cases of chemical intolerance. Environ Sci Eur 2021;33:129.ArticlePDF
- 26. Martin Perez IM, Parra Castillo DA, Ruiz de la Fuente CP, Martin Perez SE. Effectiveness of lifestyle-based approaches for adults with multiple chemical sensitivity: a systematic review. Therapeutics 2025;2(3):13.Article
- 27. Caress SM, Steinemann AC. A national population study of the prevalence of multiple chemical sensitivity. Arch Environ Health 2004;59(6):300–5.ArticlePubMed
- 28. Miller CS, Mitzel HC. Chemical sensitivity attributed to pesticide exposure versus remodeling. Arch Environ Health 1995;50(2):119–29.ArticlePubMed
- 29. Ross GH. Clinical characteristics of chemical sensitivity: an illustrative case history of asthma and MCS. Environ Health Perspect 1997;105 Suppl 2:437–41.ArticlePubMedPMC
- 30. Hauge CR, Rasmussen A, Piet J, Bonde JP, Jensen C, Sumbundu A, et al. Mindfulness-based cognitive therapy (MBCT) for multiple chemical sensitivity (MCS): results from a randomized controlled trial with 1 year follow-up. J Psychosom Res 2015;79(6):628–34.ArticlePubMed
 , Yoon-Ji Kim2
, Yoon-Ji Kim2 , Dongmug Kang1,2,3
, Dongmug Kang1,2,3 , Youngki Kim1,2,3
, Youngki Kim1,2,3 , Eun-Soo Lee1
, Eun-Soo Lee1 , Ki-Hun Kim1
, Ki-Hun Kim1 , Se-Yeong Kim1,2,3,*
, Se-Yeong Kim1,2,3,*


 KSOEM
KSOEM

 Cite
Cite