Tuberculosis infection status and risk factors among health workers: an updated systematic review
Article information
Abstract
Tuberculosis (TB) infection is a common occupational risk for health workers (HWs) and poses a threat to the patients under their care and to other HWs. Hence, the development of a prevention strategy is crucial. We conducted a study to understand the status and risk factors of TB infection among HWs. The existing literature was searched for all published reports from 1 August 2010 to 31 December 2018, related to TB among HWs according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. The inclusion criteria were: (1) study participants working in a health care facility; (2) TB cases diagnosed by medical professionals; (3) original research articles; and (4) English reports in a peer-reviewed journal. We finally included 61 studies from 642 articles searched initially. The TB infection rate in HWs was higher than that of the general population. Based on 39 studies, the prevalence of TB in HWs (tuberculin skin test positive) was 29.94%. In contrast, the global burden of latent TB infection was 23.0% (95% uncertainty interval: 20.4%–26.4%) in 2014. The risk factors of TB among HWs were aging, long duration of employment, nursing professionals, lack of Bacillus Calmette-Guerin vaccination, and low body mass index. HWs have an increased risk for TB infection, which can cause secondary infections in patients or other HWs. An effective prevention strategy must be developed to enable early diagnosis and prompt treatment.
BACKGROUND
Health workers (HWs) are routinely exposed to infected blood and body fluids at their workplaces. One third of the world population has been infected by tuberculosis (TB), including the latent TB infection (LTBI), and the highest risk of TB transmission has been reported in HWs [12]. HWs are frequently exposed to patients with undiagnosed or untreated TB at work [3]. A HW infected with TB can act as a carrier and poses a threat to other patients or fellow HWs. Thus, TB infection of HWs at work is a major concern not only in the domain of occupational health but also in public health. Despite the large number of studies on TB infection among HWs, there is a lack of clarity regarding its prevalence in different countries and its risk factors in occupational settings.
Baussano et al. [4] conducted an in-depth review of the research on the TB status among HWs. From January 2005 to July 2010, forty-three eligible studies were included. These accounted for 1,176 cases of LTBI among HWs with 49,576 person-years of follow-up and for 1,942 TB cases with > 1,010,047 person-years of follow-up. Cases of LTBI and TB among HWs were reported in 25 and 18 studies, respectively. Since then, the literature on the TB status among HWs has reported a significant increase. Thus, we conducted an updated systematic literature review for TB infection among HWs from August 2010. The aim of this review was to comprehensively and critically assess the literature regarding the intercontinental prevalence, risk factors, and the type of TB among HWs, and to gain a better understanding of TB among HWs for guiding the future research.
METHODS
Main goal and search strategy
This study systematically reviewed the TB infection status and risk factors for HWs, providing the academic evidence and a strategy to prevent HWs from TB. The main problem in the current review study was TB exposure to HWs. The TB infection status and risk factors were compared with the general population or non-HWs, outcomes were demonstrated with prevalence, incidence, or odds ratio (OR).
A systematic search was conducted on the databases of PubMed and Google Scholar for all published reports on TB among HWs according to the Preferred Reporting Items for Systematic reviews and Meta-Analyses guidelines. The search term used was: “TB” OR “tuberculosis” OR “LTBI” OR “latent tuberculosis infection” AND work* OR occupation* OR industry* OR employee*. All the relevant published articles from 1 August 2010 to 31 December 2018 were identified, as the previous review had already included the 43 studies pertaining to TB among HWs published from 1 January 2005 to 31 July 2010 [4].
Inclusion and exclusion criteria
We included all studies that satisfied the following criteria: (1) study participants working in a health care facility; (2) TB patients diagnosed by medical professionals; (3) original research articles; and (4) English reports in a peer-reviewed journal.
Studies were excluded if they were: (1) reviews, notes, case reports, commentaries, or editorials; (2) non-human focused researches; (3) articles describing only guidance of clinical treatment or other similar study designs.
Selection and organization
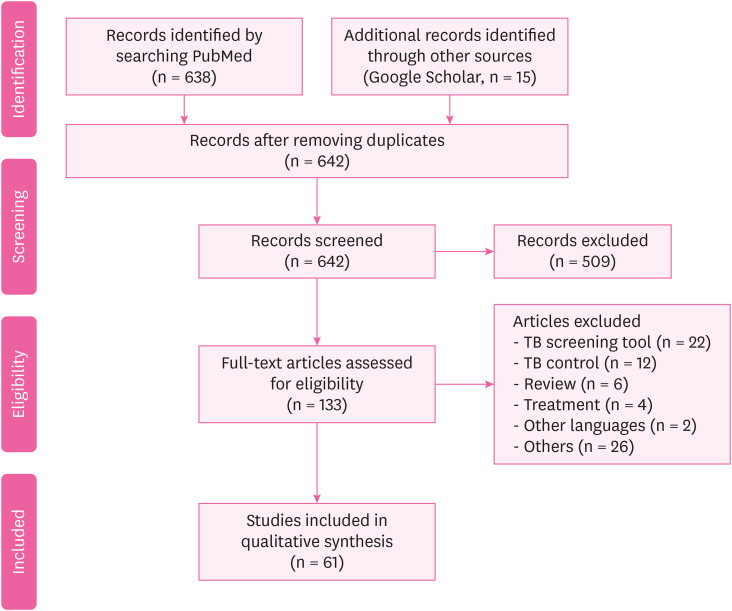
After the search, the duplicates were removed. The titles and abstracts of the remaining articles were then screened, based on the inclusion and exclusion criteria. Thereafter, the screened articles were checked for eligibility by reviewing a random sample of the included and excluded studies. Full text screening was also performed to assess the articles against the inclusion criteria to complete the process of selection and organization. A flow chart of the process is demonstrated in Fig. 1. Finally, the selected articles were organized based on them pertaining to the following two aspects; (1) the status (prevalence or incidence) of TB among HWs; and (2) the risk factors of TB among HWs.
RESULTS AND DISCUSSION
We identified a total of 638 potentially relevant citations from the PubMed data base, and 15 from the other sources (Google Scholar). The article screening and selection were conducted by three authors. Five hundreds and nine articles were excluded by the initial screening because the quality and scope of these studies did not fit those of the current study. Finally, 61 studies were included by excluding 72 studies. The included studies were divided into two categories based on them pertaining to either the status of TB among HWs or the risk factors of TB among HWs. Fig. 1 demonstrated the article selection flow.
The detailed description of the studies included in each type is as follows. The proportions and numbers of the major studies according to the geographic locations were: America 5%; Europe 23%; Asia 49%; and Africa 23%. The test methods used for the diagnosis of TB included interferon-gamma releasing assay (IGRA), 12%; tuberculin skin test (TST), 23%; survey questionnaire, 13%; and others/multiple tests, 50%. The test methods in the latter category included acid fast bacilli (AFB) stains, chest X-ray (CXR), genotyping data, and several studies used TST and IGRA together.
Both TST and QuantiFERON-TB Gold-In-Tube (QFT-GIT) are immunoassay tests used to determine whether the TB infection is latent or active. TST is considered positive if the skin over the injected portion is swollen (> 10 mm) within 48–72 hours after injecting it with Mycobacterium tuberculosis purified protein derivative. QFT-GIT is a cytokine release test, and interferon-gamma is higher than a certain level in patients with TB infection (including LTBI and active TB).
The status of TB among HWs
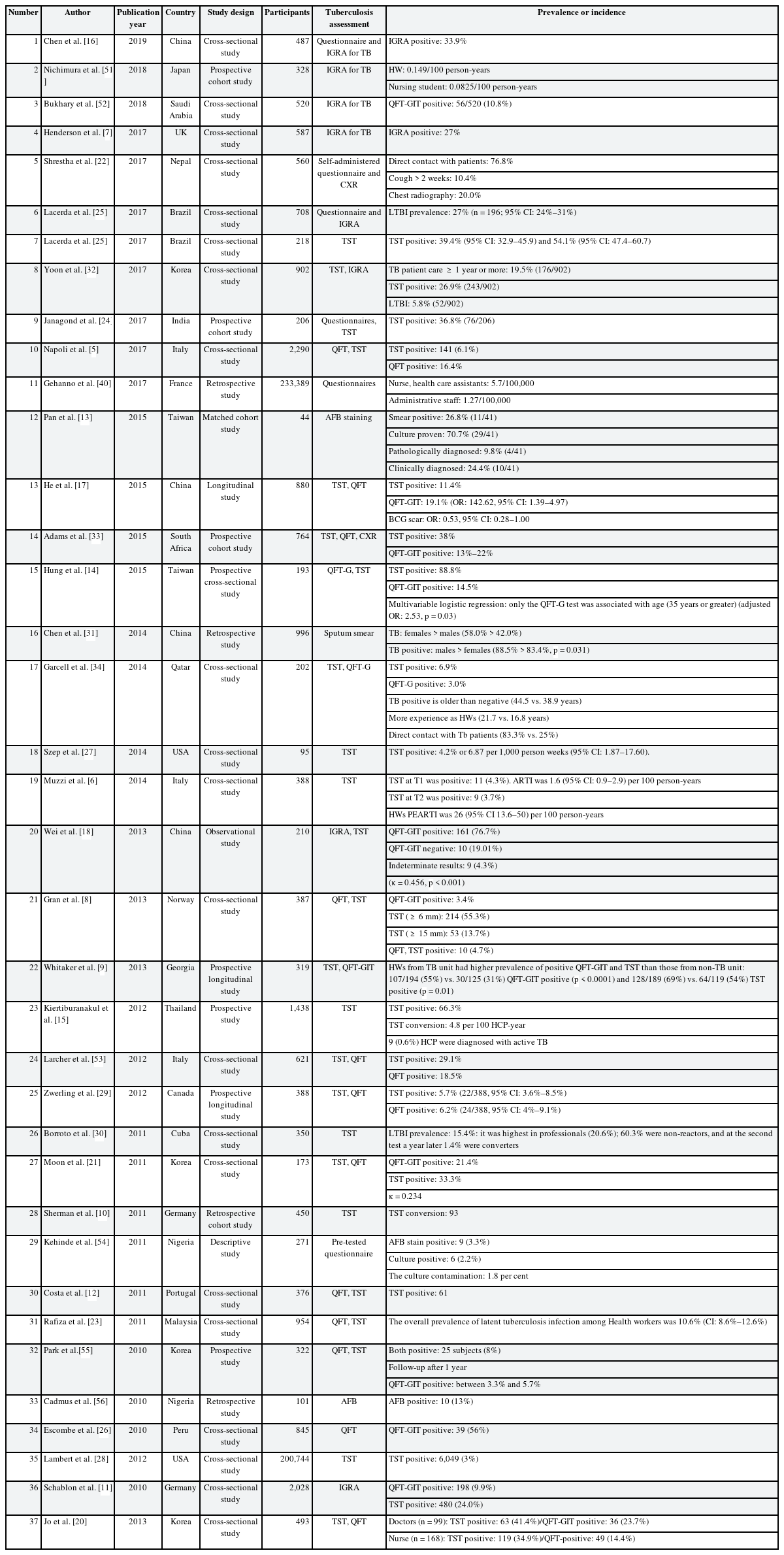
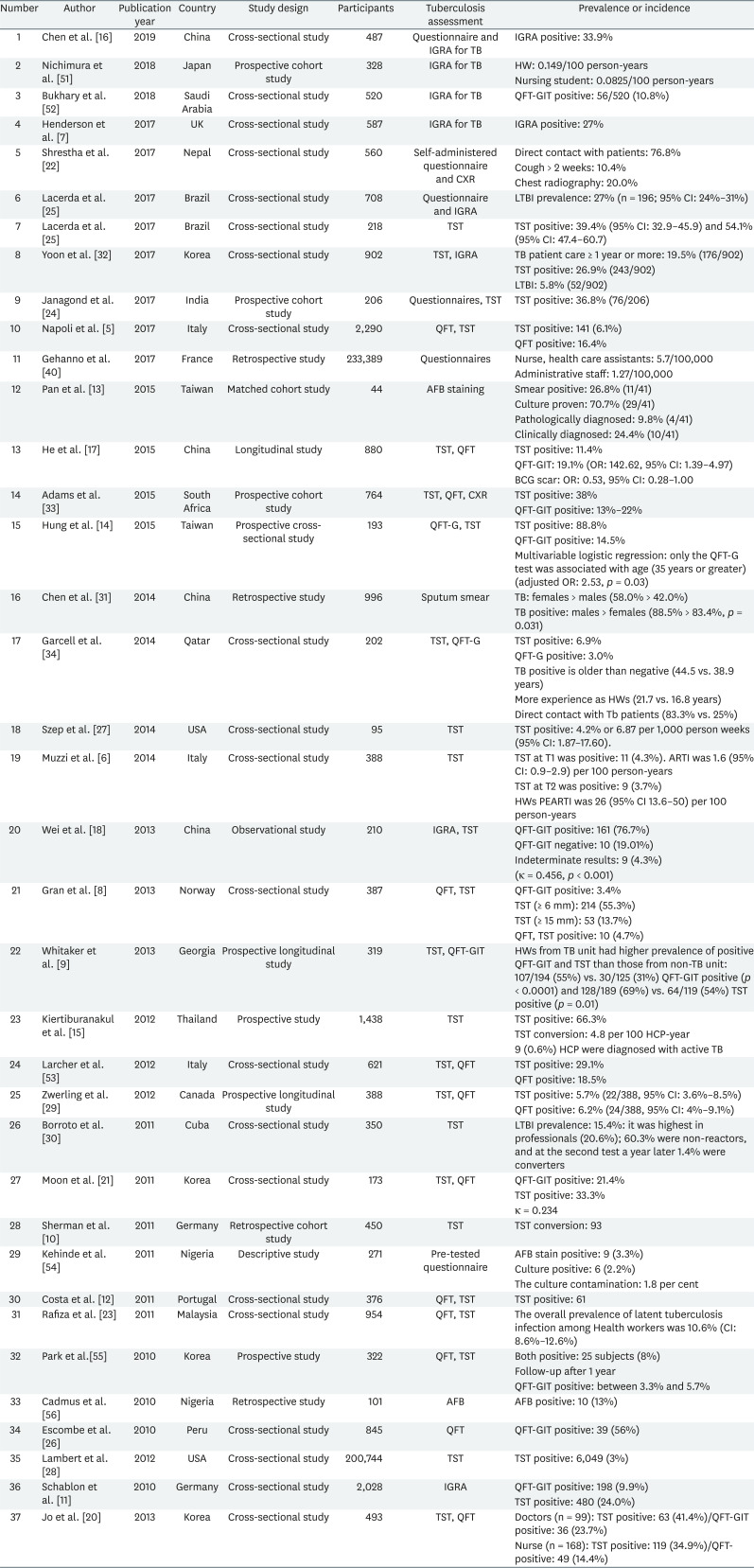
Table 1 showed the TB infection status among HWs. Based on the results of 39 studies, 29.94% of the HWs were found to be positive for TB. We compared the prevalence by the geographical location, gender, and screening test. Most of the European countries, except Italy, had about 30% positive cases. In Italy, 6.1% (TST positive) of the cases were positive in 2017 [5] compared to 4.3% in 2014 [6]. Infection rates in the other countries and the corresponding years were as follows, 27% (UK, 2017) [7]; 55.3% (Norway, 2013) [8]; QuantiFERON-TB (QFT) positive: 3.4%, TST positive: 55.3% (≥ 6 mm), 13.7% (≥ 15 mm), TST, QFT both positive: 4.7% (Georgia, 2013) [9]; 20.6% (Germany, 2011) [10]; 24.0% (Germany, 2010) [11]; and 16.2% (Portugal, 2011) [12]. Asia, like Europe, had an average of about 30% positive cases. However, Taiwan and Thailand had particularly higher rates, as follows: 24.4% (Taiwan, 2015, AFB staining) [13]; 88.8% (Taiwan, 2015, TST) [14]; 66.3% (Thailand, 2012, AFB staining) [15]. In China, the rates decreased slightly in 2015 (11.4%) [16] but again increased to 33.9% in 2019 [17], similar to the trend seen in 2013 (33.6%) [18]. In Korea, the infection rate showed a decreasing trend: 26.9% (2017) [19]; 41.4% (2013) [20]; and 33.3% (2011) [21]. Nepal and Malaysia had lower prevalence rates of 10.4% (2017) [22] and 10.6% (2011), respectively [23], while India's rate in 2017 was 36.8% [24]. In the American continent, the reported prevalence was 39.4% (Brazil, 2017) [25]; 56% (Peru, 2010) [26]; 4.2% (USA, 2014) [27]; 3% (USA, 2012) [28]; 5.7% (Canada, 2012) [29]; and 15.4% (Cuba, 2011) [30].
There were 4 articles reported the prevalence of LTBI from thirty-eight articles about TB infection status. The status of LTBI was different by countries, in Korea 5.8% [19]; in Malaysia 10.6% [23]; in Cuba 15.4% [30]; in Brazil 27.0% [25].
Higher TB infection rates were identified in women (women, 58.0% and men, 42.0%) [31]. The positive rates with the QFT-GIT test (5.8%, 52/902) were lower than those with the TST (26.9%, 243/902) for LTBI [32]. Several other studies showed a similar trend as follows: TST: 38% and QFT: 13%–22% [33]; TST: 88.8% and QFT: 14.5% [14]; TST: 6.9% and QFT: 3.0% [34]; and TST: 55.3% and QFT: 3.4% [8].
The risk factors of TB among HWs
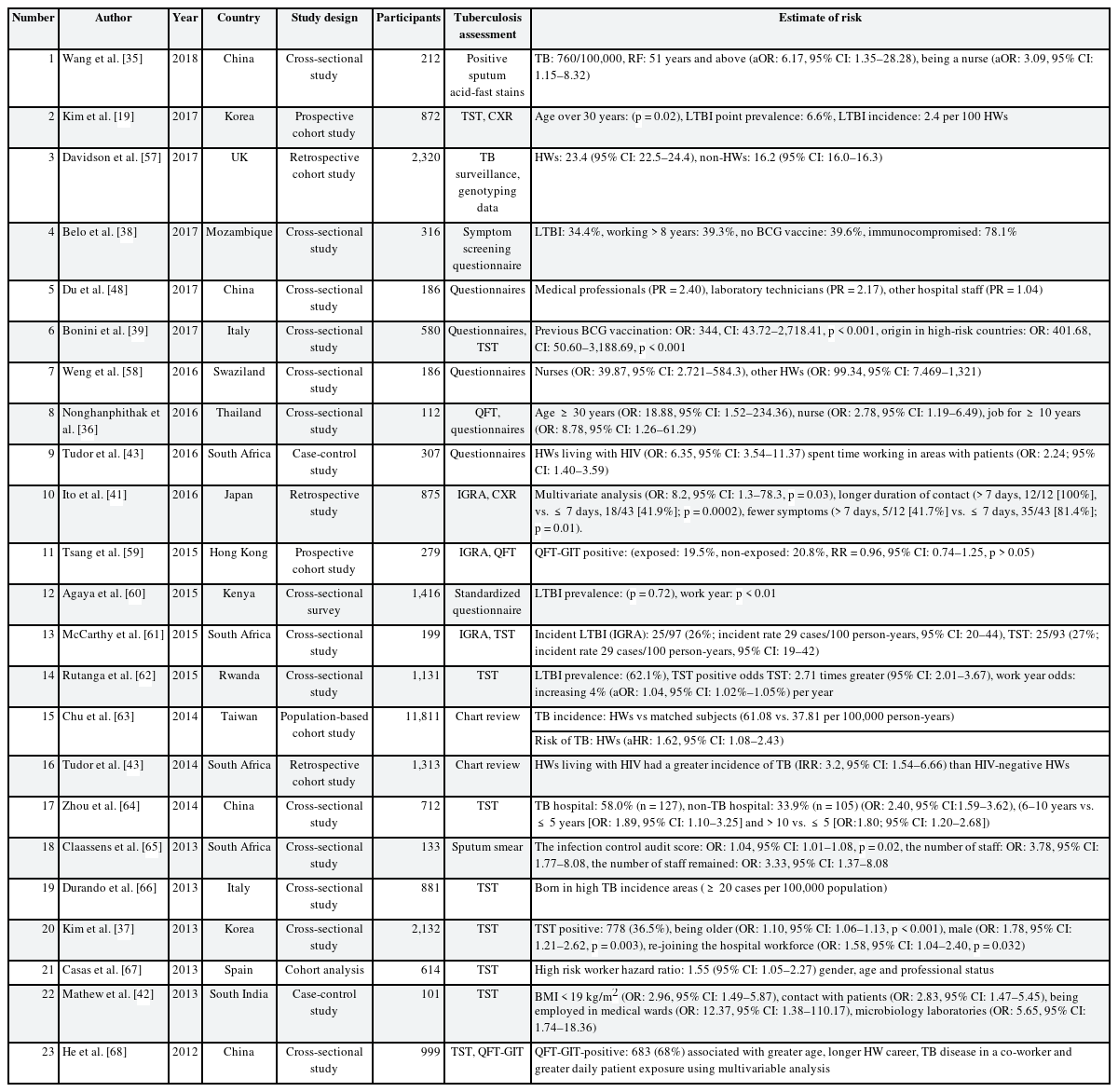
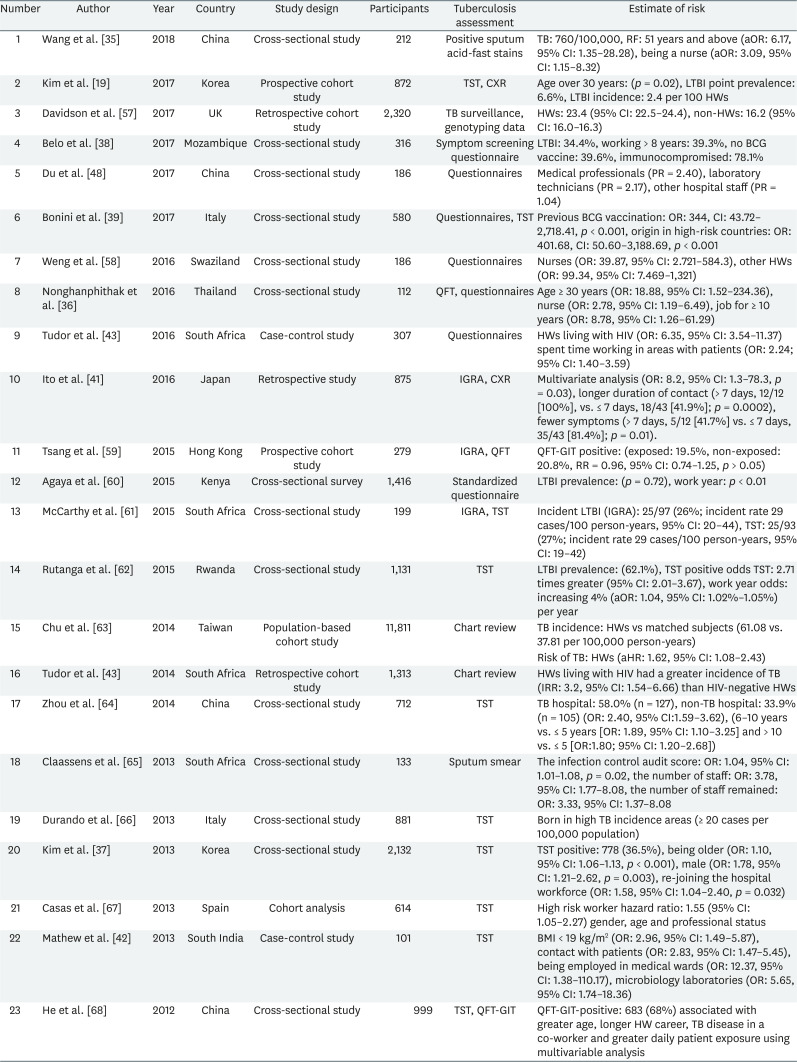
Table 2 showed TB infection related risk factors status among HWs. Risk factors such as old age, long work-duration, lack of Bacillus Calmette-Guerin (BCG) vaccination, nursing professionals, TB contacts, and lower body mass index (BMI) were identified. Patients older than 51 years showed a higher TB infection rate (OR: 6.17, 95% confidence interval [CI]: 1.35–28.28) [35]. Age > 30 years was associated with a positive TST (p = 0.02 [19]; age ≥ 30 years, OR: 18.88, 95% CI: 1.52–234.36 [36]; being older, OR: 1.10, 95% CI: 1.06–1.13, p < 0.001 [37]).
The longer the number of work years of the HWs was, the higher the TB infection rate was. For instance, 39.3% HWs with work years > 8 years were TB positive [38], work years ≥ 10 years were also associated with a higher risk [36]. TB infection rates were also higher in the HWs (39.6%) [38] who did not receive the BCG vaccination with a study reporting an OR of 344 (CI: 43.72–2,718.41, p < 0.00001) [39].
Nurses had a higher OR than the other HWs (being a nurse, OR: 3.09, 95% CI: 1.15–8.32 [35]; nurses OR: 39.87, 95% CI: 2.721–584.3 and other HWs, OR: 99.34, 95% CI: 7.469–1,321 [40]; and nurses, OR: 2.78, 95% CI: 1.19–6.49 [36]).
There was an increased risk for TB with longer working durations (> 7 days, 12/12 [100%] vs. ≤ 7 days, 18/43 [41.9%]; p = 0.0002) [41] and direct contact with patients (OR: 2.83, 95% CI: 1.47–5.45) [42]. The infection was also observed more frequently in underweight HWs (BMI < 18.5, OR: 2.96, 95% CI: 1.49–5.87) [42], or in HWs with human immunodeficiency virus (OR: 6.35, 95% CI: 3.54–11.37) [43].
Discussion
Our results showed that the risk for TB among HWs was higher than that of the general population worldwide. Furthermore, differences were found in this association according to the geographical location. Risk factors for LTBI included old age, long work-duration, nursing professionals, lack of BCG vaccination, and lower BMI.
Overall, 29.94% of the HWs were found to be TB test positive. The rates were lower in Italy (4%–6%) and higher in Thailand and Taiwan. However, there was no significant difference between the TB infection rate of HWs in Taiwan and that of the general population. This suggests that HWs in countries with intermediate TB burden, face a similar risk of exposure to TB as the community, and that appropriate personal protection equipment and good infection control practices within a hospital effectively mitigates the transmission of TB in healthcare settings. Further, HWs who did not report an exposure to TB may have been unknowingly exposed to it either within the hospital or in the community [14]. However, these results must be interpreted with caution because TB infection status could be different by TB diagnosis tools.
Studies from different geographical locations were examined. As TB prevalence is still high in Asia and continental Europe, the research in these regions appeared to be more active than in the Americas. In France, the incidence of TB among the nurses and paramedics was not higher than the general public. This probably reflects the effectiveness of the measures implemented over the last decade to prevent the transmission of TB in the health care industry, as well as an overall decline of TB in the general population.
In our review, the prevalence of TB positive in doctors and nurses were high, which was similar to the existing literature. In terms of the occupation, TB prevalence was highest in the nurses, followed by doctors. This was probably due to their close proximity with the patients, particularly those with severe infections requiring high-risk investigations, such as bronchoscopy [35].
For the diagnosis, most of the studies used IGRA and TST together, while some others used either IGRA or TST, individually. Others employed questionnaires alone or along with the test. Additionally, AFB stains, CXR, and genotyping data were also used. TST and IGRA are currently used as the basis for LTBI screening and have different guidelines in different countries. Two or more screening methods are recommended for TB screening in adult and immunocompromised populations. Sometimes one test can provide an advantage over the other. For example, the IGRA is preferred over TST in groups where BCG is administered after infancy or at multiple times. However, none of the present LTBI tests fulfil the requirement of early diagnosis of TB. In a study comparing the results of IGRA and TST conducted in Saudi Arabia for medical workers, the following results were found. The disagreements between the two tests were relatively high (kappa co-efficient = 0.312 ± 0.026, p < 0.00001) as TST positive/QFT negative discordance was 54.8% while TST negative/QFT positive discordance was 15.7% [44]. Therefore, supplementary testing methods such as CXR, AFB stains and questionnaires should be used.
Various risk factors were identified. Aging, diabetes mellitus and low BMI, are known factors of immunological imbalance associated with TB development in the general population [45]. The reason for higher TB infection in older HWs and longer work years was due to an increased contact with TB patients in addition to the aging and decline in the immunity [35]. Although a low BMI is associated with active TB, it can be interpreted as a consequence of TB rather than as a risk factor [4246].
According to the World Health Organization (WHO) guidelines, strict clinical observation and close monitoring for the development of active TB for at least two years is preferred over the provision of preventive treatment for contacts of multidrug-resistant-TB cases. In cases where preventive therapy is considered for such individuals, the program should ensure that the necessary resources are available to provide quality-assured drug susceptibility testing and the necessary medications, and to closely monitor for any harm, breakthrough disease, and acquired resistance [47].
In many of the countries, despite the increasing risk of TB infection in HWs, the quality of occupational health and safety, and infection prevention and control have not improved significantly. Evidence-based investment of effort is required for the effective risk reduction for HWs [4849].
The Republic of Korea has a moderate TB prevalence (85–110/100,000 population per year) and BCG vaccination is recommended at birth [37]. Until 1997, the BCG vaccine was given at birth and repeated at the age of 12 or 13 years if the child proved to be a TST non-responder. However, revaccination was stopped in 1997 according to the WHO recommendation in 1995 due to the lack of evidence [50].
Further research is needed to assess the change in regional surveillance systems in different countries, depending on the development of respective national health conditions. We also need a country- and age-wise evaluation of the effectiveness of the BCG vaccination.
In conclusion, this study showed that HWs were at higher risk for developing TB. It further emphasized on the need for sound TB infection control measures, including early diagnosis and prompt treatment, for its effective prevention in HWs and the general public.
Notes
Competing interests: The authors declare that they have no competing interest.
Author Contributions:
Conceptualization: Kang SK.
Formal analysis: Lee S.
Investigation: Lee S, Lee W.
Methodology: Lee W, Lee S.
Validation: Kang SK, Lee W.
Writing - original draft: Lee S, Lee W, Kang SK.
Writing - review & editing: Lee S, Lee W, Kang SK.
Abbreviations
AFB
acid fast bacilli
aOR
adjusted odds ratio
ARTI
annual rate of tuberculosis infection
BCG
Bacillus Calmette-Guerin
BMI
body mass index
CI
confidence interval
CXR
chest X-ray
GIT
QuantiFERON-TB Gold-In-Tube
HIV
human immunodeficiency virus
HR
hazard ratio
HW
health worker
IGRA
interferon-gamma release assay
IRR
Incidence rate ratio
LTBI
latent tuberculosis infection
OR
odds ratio
PR
prevalence ratio
QFT
QuantiFERON-TB
QTF-PEARTI
post-exposure rate of tuberculosis infection
RF
risk factor
RR
risk ratio
TB
tuberculosis
TST
tuberculin skin test
WHO
World Health Organization